Host recognition and resistance reactions in the interaction of the parasitic weed Orobanche cumana and cultivated sunflower
Anna Krupp (PhD Defence 2020)1)
Supervisors: Otmar Spring1), Anne Heller1), Joachim Sauerborn2)
1) University of Hohenheim, Institute of Botany
2) University of Hohenheim, Institute for Plant Production and Agroecology
Challenges:
Parasitic plants are a serious problem for the cultivation of some important crops, particularly in arid areas. Thus, Striga endangers grain crop production in Africa, various Orobanche and Phelipanche species reduce the yield of legumes and tomato in the Near East and more recently sunflower broomrape (Orobanche cumana Wallr.) rapidly expanded in the summer-dry regions of Europe, Africa and Asia for which sunflower (Helianthus annuus L.) is amongst the most important oil crops.
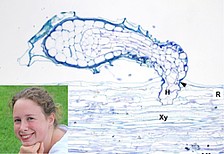
O. cumana produces numerous tiny seeds that stay viable in soil for many years until signals from host roots induce germination and attachment to the host. After penetrating the host root and forming a connection to its vascular system, the parasite depends entirely on its host for water supply and nutrients, thus causing severe yield losses. The control of this parasitic weed is extremely difficult, because control measures may also affect the crop plant. Gaining more knowledge about parasitizing and resistance mechanisms is crucial to develop effective control strategies that can help to ensure food production especially in areas which already suffer from limited water supply.
O. cumana specifically parasitizes sunflower and does not accept other hosts. There are defined pathotypes of O. cumana that have been identified by breeding host genotypes with specific resistance genes. This makes this host-parasite-combination an ideal model system for the investigation of interaction processes. Moreover, major parts of the genome of sunflower are sequenced and the host is easily cultivated under laboratory conditions all year long.
Objectives:
In my PhD work I will focus on two main parts of this interaction: (I.) The host finding mechanism including chemotropism, host root recognition and attachment, and (II.) haustorium growth regulation and its mechanisms to reach the vascular system without causing defense reactions of the host. Special emphasis will be put on resistance reactions of the host, thus deducing strategies and goals for plant breeding and cultivation techniques that can reduce parasitic plant infection and therefore minimize yield losses.
Methods:
Microscopy (to understand the anatomic interaction of host and parasite and resistance reactions), bioassay-guided chromatography (to find new bioactive substances that have an influence on germination and chemotropism) and molecular genetic techniques for the identification of genes involved in pathogenicity and resistance. Experiments comprising different cultivation techniques such as root chambers, where host-parasite-interaction can be directly monitored, and hydroculture for root exudate collection will be conducted. Samples from the root chambers representing specific developmental stages of the interaction will be collected for microscopic studies and molecular-based studies, whereas root exudates will be used for bioassay-guided chromatography to identify substances with an effect on germination, chemotropism, and host recognition.
Expected results are:
- Identification of new bioactive substances involved in the host recognition of O. cumana / H. annuus (germination stimulants/inhibitors, chemotropism-related compounds, signal molecules released during root attachment)
- Identification of cellular processes leading to the success or failure in penetrating the host's vascular system by comparing susceptible and resistant host-parasite-combinations
- Detection of defense reactions involved in the resistance of sunflower
- Detection of putative mechanisms of broomrape to suppress the host's resistance reaction
- Deduce strategies and goals for plant breeding and cultivation techniques that can reduce broomrape infection and therefore reduce yield loss in arid areas of sunflower cultivation
- Get information from this model system that can be transferred to other crop systems suffering from parasitic weeds of other Orobanche, Phelipanche and Striga species
Publications
- Krupp, A., Rücker, E., Heller A. & Spring, O., 2014. Seed structure characteristics of Orobanche cumana populations. Helia, DOI: 10.1515/helia-2014-0034
- Güney A., Zimmermann R., Krupp A. & Haas K, 2016. Needle characteristics of Lebanon cedar (Cedrus libani A. Rich.): degradation of epicuticular waxes and decrease of photosynthetic rates with increasing needle age, Turkish Journal of Agriculture and Forestry, 40, 386-396
- Krupp, A., Heller, A. and Spring O.,2019. Development of phloem connection between the parasitic plant Orobanche cumana and its host sunflower, Protoplasma 256(61)